Chemische Physik fern des Gleichgewichts
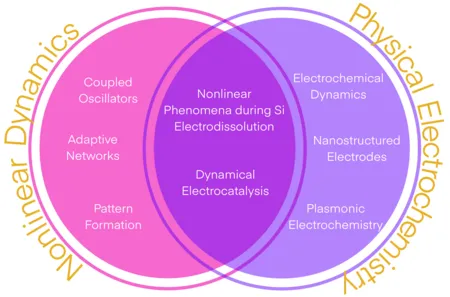
Our research is centered around the dynamics of non-equilibrium systems. We explore their behavior experimentally and theoretically. Our experimental studies focus on the reaction dynamics of electrochemical systems, such as the CO2 reduction and the hydrogen evolution reaction. The prototypical experimental model system for the study of pattern formation and emergent behavior is the electrodissolution of Si. Our theoretical work deals with the universal behavior in certain classes of non-equilibrium systems. Most of the theoretical studies are motivated by our experimental results and address fundamental laws that govern behavior in complex systems. Examples include collective dynamics of coupled oscillators and pattern formation in reaction-diffusion systems.
Latest group news and publications can be found here.
Nonlinear Dynamics
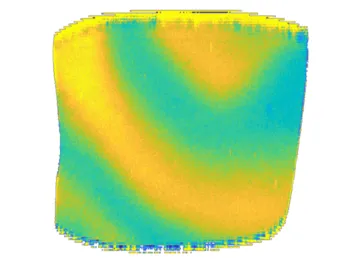
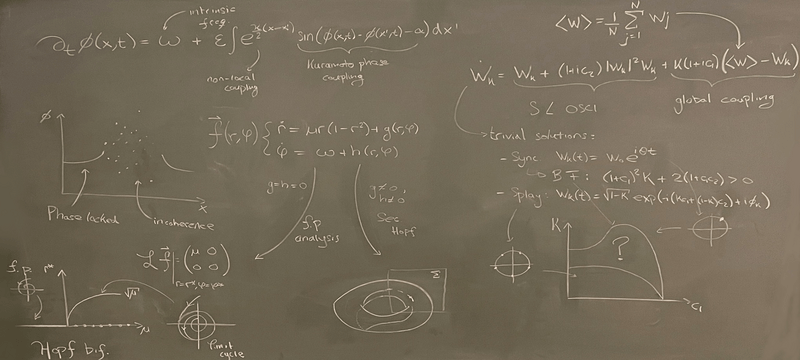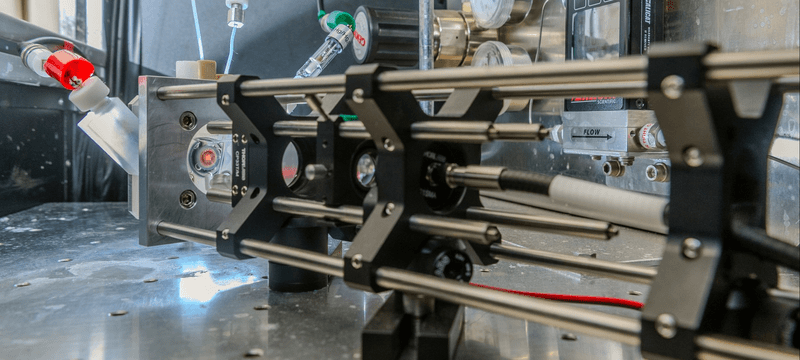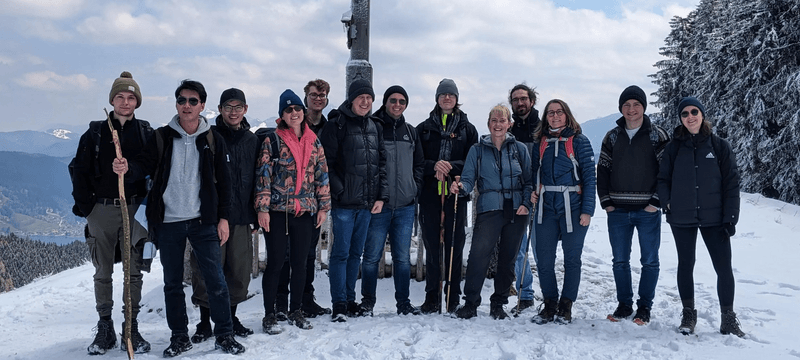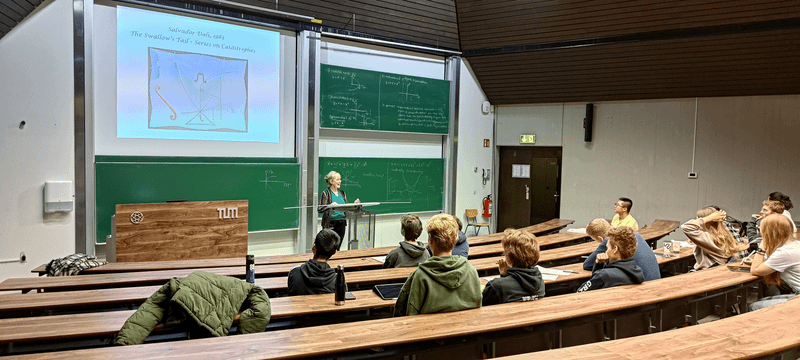
Nonlinear dynamics deals with emergent behavior, i.e. all kinds of phenomena in dynamical systems that are ‘more than the sum’. This comprises the spontaneous formation of patterns from originally uniform states as well as the collective dynamics of an ensemble of coupled oscillators. We investigate synchronization patterns in systems of coupled oscillators and oscillatory media both experimentally and theoretically.
Our research topics in the field of Nonlinear Dynamics
Physical Electrochemistry
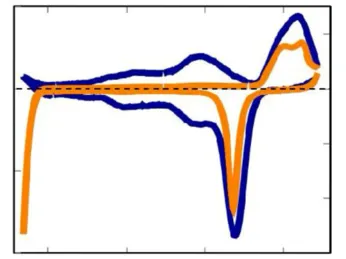
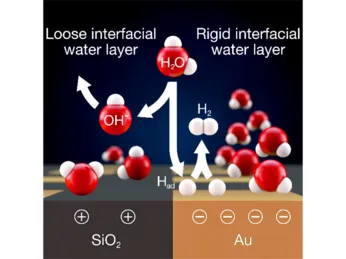
Physical Electrochemistry covers all fundamental aspects of the structure and the dynamics of interfaces between an electronic and an ionic conductor, from the molecular to the macroscopic scale. We consider electrified interfaces between a solid and a liquid phase. Particular attention is paid to the effects of spatially or temporally varying parameters on the interfacial properties and reactivity of the system as well as on conditions and mechanics that lead to the spontaneous emergence of spatial and temporal structures. This includes reactions on nanostructured electrodes, reactions under time-periodic forcing, and reactions that exhibit self-organized reactivity patterns at the electrode surface. The reactions we are investigating include the CO2 reduction reaction, the hydrogen evolution reaction and Si electrooxidation. In addition to the study of fundamental mechanisms governing the reaction dynamics, we develop experimental techniques for the sensitive measurement of changes in the structure or composition of the interfacial layer, e.g., using plasmonically active electrodes.
Our research topics in the field of Physical Electrochemistry